 Same formula, same conversion factor – yet chemical reactions are much less violent than nuclear explosions. The same formula applies: The mass difference, multiplied by c 2, gives the energy set free during the chemical reaction. The same is true for the chemical reactions involving spontaneous oxydation – in other words: burning. To pick an example: When hydrogen and oxygen explosively combine to make water, the sum of the rest masses of the initial hydrogen and oxygen atoms is just a little bit less than the sum of the rest masses of the resulting water molecules. Different process, same calculation: For chemical reactions, there are tiny mass differences as well. So is it, after all, true that Einstein’s formula explains the power of the nuclear bomb – and that the large conversion factor c 2 is responsible for the immense amounts of energy released? Binding energies: nuclei vs. Here’s where E=mc 2 comes into play: This mass difference corresponds to the energy set free during nuclear fission. For example, the rest mass of a nucleus of uranium-235 is slightly larger than the combined rest masses of the nuclear fragments into which it splits during nuclear fission. Studying the masses of different types of atomic nuclei, you will find that in nuclear fission – the process that powers an ordinary atomic bomb -, some “nuclear rest energy” or “nuclear rest mass” is transformed into other forms of energy. For instance, when a particle and its anti-particle annihilate and vanish in a puff of electromagnetic radiation, comparatively little matter is transformed into rather a lot radiation. This high concentration is important for processes where rest energy (or, equivalently, rest mass) is converted to more common forms of energy. 
Also, this rest energy is about a hundred times larger than the radiation energy of a high-energy X-ray photon. For example: If you use a television tube to accelerate an electron to 20,000 kilometres per second, the kinetic energy gained is still only about five hundred times smaller than the electron’s rest energy. This is called the particle’s rest energy, and it is related to the particle’s rest mass asĬompared with other types of energy, rest energy is very much concentrated. However, in relativity, the definitions of the different species of energy are a bit different and, most importantly, there is a completely new type of energy: even if a particle is neither moving nor part of a bound system, it has an associated energy, simply because of its mass. This conservation of energy holds not only in classical physics, but also in special relativity. Energy can be transformed from one variety into another, but it can neither vanish nor be created from nothing. In these transformations, the total sum of all the different kinds of energy – the total energy – is constant over time. For instance, you can increase a body’s temperature (and thus its thermal energy) by letting it absorb electromagnetic radiation energy. The reason is the possibility of transformations between the different energy forms. Yet all these different definitions can be viewed as facets of a single physical quantity, energy. Already in classical, pre-Einstein physics, the concept of “energy” comprises a plethora of sub-definitions for different sorts of energy, sub-definitions like those for the kinetic energy associated with any moving body, the energy of electromagnetic radiation, thermal energy or the binding energy that needs to be taken into account whenever there is a force holding together two objects to form a composite object. 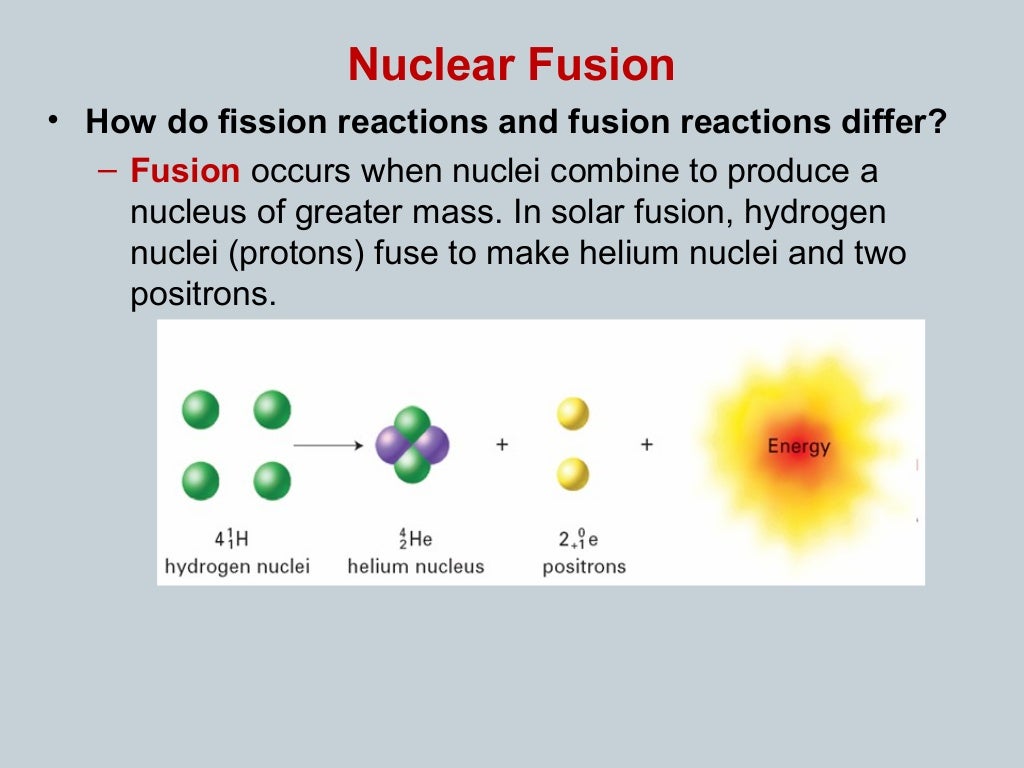 
It is intimately connected with the fact that there are different kinds of energy. The context in which “transformation of mass into energy” does make sense is a bit different. In that context, it makes no sense to talk about the “transformation of mass into energy” – where there’s one, there’s the other. Once the mass is known, so is the energy, and vice versa. Whenever a system has an energy E, it automatically has the relativistic mass m=E/c 2 whenever a system has the mass m, you need to assign it an energy E=mc 2. Equivalence or transformation?įor Einstein, mass (more precisely: relativistic mass the property that determines how difficult it is to change a body’s speed or its direction of motion) and energy are simply two different names for one and the same physical quantity. Let’s have a look at what Einstein really did say about the relation between mass and energy. Ten seconds after the ignition of the first atomic bomb, New Mexico, Jīut first things first. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
